Phylogeny & Infectious Diseases
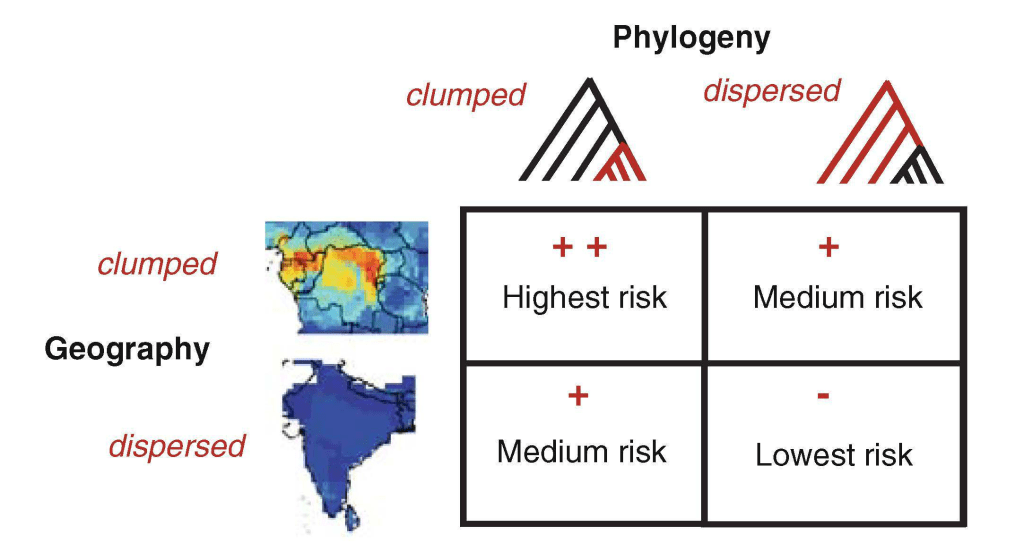
Representation of the interaction between phylogeny and geography that may determine the frequency of host shifts within host communities. In the maps, warmer colors represent areas with many overlapping species ranges (geographically clumped distributions). In the phylogeny, red branches represent samples of closely related taxa (phylogenetically clumped). More…
Infectious diseases cause 1 in 5 human deaths and are responsible for 51% of years of life lost globally. In a study on the global distribution of human diseases, the best predictor of disease richness is the number of animal species within a region (Dunn, Davies et al. 2010 PRS 277:2587). In part, this correlation may be explained by diseases shifting from wildlife to humans. Humans share parasite communities with wild animals in proportion to their phylogenetic relatedness (Davies & Pedersen 2004 PRS 275:1695). We might, therefore, predict hotspots of parasite host-shifts to humans, and thus centres of zoonotic disease emergence, to be located in regions of high primate diversity (Pedersen & Davies 2009 EcoHealth 6:496).
Identifying the potential for transmission of parasites among hosts will be important for directing surveillance of animal parasites before they successfully emerge in humans, and increasing the efficacy of programs for the control and management of zoonotic diseases (Farrell, Berrang-Ford & Davies 2013 Environ. Res Lett 8:015036).
In a study of African primate communities, our lab has revealed that true parasite diversity is likely severely under-estimated, and that even for relatively well-studied parasites there might be considerable cryptic species diversity (Ghai et al. 2014 PLoS Negl Trop Dis 8:e2641). Many of these species might be shared between humans and wild primates, but such parasite sharing has remained largely undocumented (Ghai et al. 2014 PLoS Negl Trop Dis 8:e3256). It should not be forgotten, however, that pathogens are important components of healthy ecosystems; pathogens can promote genetic diversity in their hosts, facilitate species coexistence within communities, and provide natural biological control of weeds and pest species. As we lose host species through extinction we also lose the parasitic diversity that is associated with them. In a surprising result, we showed that it is the generalist pathogens that might be lost first, and not the host-specialists, as previously assumed (Farrell et al. 2015 J Anim Ecol 84:978). Because generalist pathogens, by definition, are shared with more hosts, their loss may have larger (but unknown) ecosystem consequences.
Since 2013, lab members have been involved in an NSF funded Research Coordination Network on the Macroecology of Infectious Disease which is working towards a new synthesis of infectious disease biology that spans across ecological and evolutionary scales (see Stephens et al. 2016 Ecol Lett 19:1159).

Marie Toorians is currently working on Bovine tuberculosis

Isidora Silva-Valderrama is currently working on the Botryosphaeriaceae fungal family in Grapevines